A Vision for the Future of Kidney Replacement Therapy

This blog post is based upon the approach of Working Group 3 “Groundbreaking Innovation” of the European Kidney Health Alliance (EKHA), as well as upon the content of two recently published open access invited reviews in the Clinical Kidney Journal and the American Journal of Kidney Diseases. If the blog interests you, we encourage you to also read these background articles.
A vision from the European Society for Artificial Organs (ESAO) Conference
Working on realizing new Kidney Replacement Therapies (KRTs) now and then brings me to conferences where innovators meet. The European Society for Artificial Organs (ESAO) is one of those places-to-be for me. At a wonderful ESAO conference held in Bergamo (Italy) I received a vision that I’d like to share with you.
The conference was held in an ancient monastery with beautiful frescos on the wall. And while watching three of these frescos next to each other, it was as if these three people started speaking to me…

Since the first ever clinically successful hemodialysis treatment in the world (1945, by Dr. Kolff in Kampen, the Netherlands), blood has been led out of the body, filtered in a big machine and then brought back into the body again. But our kidneys do their task inside the body. Wouldn’t it thus be great if we could miniaturize hemodialysis to such a degree that it might become implantable (or at least partly implantable)? This thought may be less weird than it looks like at first sight. Let me explain.
A Roadmap for Kidney Replacement Therapies (KRTs)
In 2018, the Kidney Health Initiative published an innovation roadmap for kidney replacement therapies, which can be freely downloaded here: KHI_RRT_Roadmap1.0_FINAL_102318_web.pdf
This roadmap describes several stepwise trajectories towards an artificial kidney, which can grossly be distinguished in three approaches:
Via purely technical means (devices)
Fully biological pathway (xenotransplant, lab cultivated or 3D-printed kidney)
Hybrid approach using a mixture of technology and biology
As a technologist, I am not an expert at all on the fully biological approach, hence I focus on the techno and the bio/techno hybrid approaches here (while acknowledging that exciting progress is made in xenotransplantation).
The KHI roadmap steps can be summarized as “first make KRT small, so that it becomes portable or wearable, and then make it implantable” as depicted by the cartoons in figure 2:
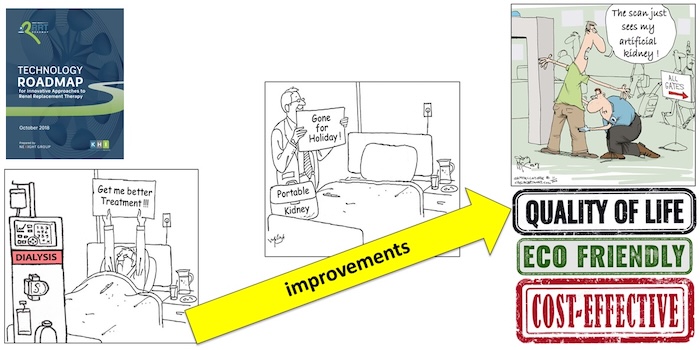
A previous blog post by Dori Schatell, shows that hopeful movements are ongoing towards making home dialysis systems smaller. But, they only become portable in a practical sense when they meet the criteria for hand luggage for air travel. That point now has been reached, as described in a recent review (freely downloadable here). This very high degree of miniaturization is enabled by regenerating the dialysate in a closed loop, instead of throwing it away after a single passage through the dialyzer.
A traditional “single-pass” hemodialysis machine uses a high dialysate volume per treatment. While there are now miniaturized single-pass machines on the market (which e.g. use low-flow approaches) these are approaching the engineering limits for further miniaturization (see Fig. 3a & 3b).
In contrast, a dialysate-regenerating hemodialysis machine can use 10 to 24 times as little dialysate (thus also consuming far less electricity to warm-up that dialysate), which enables engineers to further miniaturize (see Fig. 3c & 3d). This approach makes it possible to pack a hemodialysis machine into hand luggage for air travel.
But that still doesn’t meet the vision of figure 1…
Implantable Dialysis Filters
This vision, however, can become possible when the dialysis filter is made implantable (a vision formulated by the US-based “Kidney Project”, which already won multiple KidneyX Prizes). In that case, the heart forms the blood pump (just as with our kidneys) and the blood stays in the body while it is being cleaned, while only extracorporeal connections would be needed for the dialysate circuit (see Fig. 3e). This still would be hemodialysis, but it would mean a huge step forward, as the need for an extracorporeal blood circuit would disappear! Such an approach would make frequent nocturnal hemodialysis accessible for patients who depend on hemodialysis to survive. It would be much easier to do-it-yourself and thus also much cheaper.
Even further into the future, an additional bioreactor might be added so no extracorporeal connection would be needed at all, and concentrated artificial urine would leave the body via the natural route.
These approaches lean on solid science and are already being worked upon by research teams in both the USA (“the Kidney Project”) and in Europe (KIDNEW and NXTGEN Hightech). The biggest technological enabler is using chip-technology to make dialysis filters with an unprecedented pore diameter uniformity and very high porosity per square mm. Yet, the biggest challenge is how to ensure that such an implanted device will remain functional (without clogging) for several years (as replacement will only be possible by surgery).
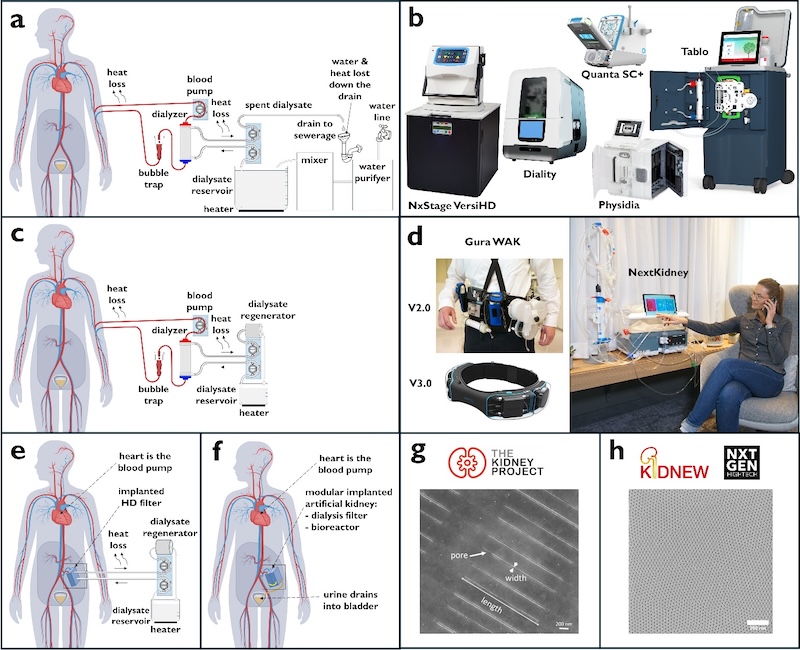
(A) Single-pass HD block diagram.
(B) Some examples of existing transportable single-pass HD devices (not exhaustive; photos from manufacturers’ websites). No commercial single-pass HD devices were found that meet International Air Transport Association carry-on baggage guidelines.
(C) Dialysate-regenerating HD block diagram.
(D) Some examples of dialysate-regenerating HD devices that reached human clinical trials and meet International Air Transport Association carry-on requirements for air travel.
(E) Partly implantable HD principle. The natural heart forms the blood pump, and an extracorporeal dialysate-regenerating device can be connected to perform dialysis.
(F) Fully implantable technological/biological hybrid artificial kidney principle with a nanoporous (e.g. silicon wafer–based) filter and a bioreactor. The natural heart also forms the blood pump in this case.
(G) Silicon nanoporous membrane of The Kidney Project with patterned slit pores.
(H) Silicon nanoporous membrane pore patterning technology as applied by imec in the KIDNEW and NXTGEN HighTech projects. (Scale bars in G and H: 200 nm). Reproduced from [REF].
A Roadmap for Kidney Replacement Therapies (KRTs): Timing is Everything
A logical and legitimate question that is commonly heard upon presenting a future vision, is: “when will such (partially) implantable devices become clinically available?” Typically, this question is asked to the innovators working on these devices. But the answer mainly depends on the decisions (and actions) of policymakers. This can be explained by the so-called “triangle of project management” which is a core part of every engineering education (see Fig. 4).
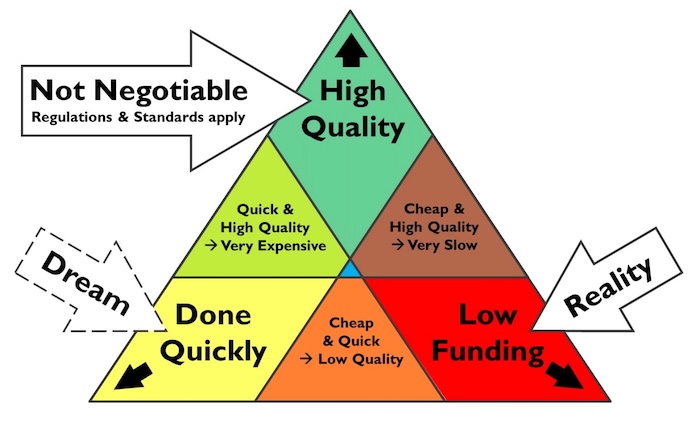
Note that the budget needed to realize a clinical product that meets at least Fig. 3e, is the money for building about 200 km of 4-lane highway. That is a very substantial sum of money, but within the reach of any developed nation (if their government is determined about the need). Unfortunately, no single nation holds all the top-experts needed to realize the project (as they live in various nations across the world). Hence, on June 24th 2025, there will be a meeting in the city of Kampen, the Netherlands (the birthplace of hemodialysis) to facilitate direct discussions between innovators, patients health-care professionals, entrepreneurs and policy makers. If several nations contribute funding and expertise, the speed of innovation can soar. So, we seem to need a “Kennedy moment.”


Comments